Ursula Bentrup
Leibniz-Institut für Katalyse e.V. an der Universität Rostock (LIKAT Rostock), Albert-Einstein-Str. 29a, 18059 Rostock, Germany
Introduction
In situ spectroscopy is the only analytical approach with which one can derive reliable information on mechanisms and the role of intermediates in chemical reactions as well as on structure–reactivity relationships in catalysis. This is also the case for the real-time monitoring of complex reactions in the liquid phase, including the precipitation of solids by, for example, the preparation of mixed oxide catalyst precursors. Considering the optical spectroscopies, infrared (IR), Raman and ultraviolet/visible (UV/vis), it has to be considered that each spectroscopic method has its distinct advantages and disadvantages. Hence, coupling of these methods provides the possibility of obtaining a more complete picture due to the increased amount of incoming information.1,2,3
Raman and IR spectroscopy provide complementary information. While IR spectroscopy enables the identification of characteristic functional groups by inspecting the range between 4000 cm–1 and 700 cm–1, bands of metal–non-metal vibrations appearing in the low frequency range (1000–200 cm–1) can be observed well with Raman spectroscopy. UV/vis spectroscopy gives less distinct information about the molecular structure. The bands observable in the UV/vis spectra of organic molecules are prominent for chromophores or indicate charge transfer between metal and ligands as well as changes of oxidation state in metal complexes. For this reason, UV/vis spectroscopy is a powerful tool to study changes in complex molecules influenced by distinct interactions with ligands and solvent molecules. Furthermore, UV/vis spectroscopic measurements in reflection mode allow the direct monitoring of precipitate formation and dissolution.
In the following, the benefit of method coupling will be demonstrated on two selected examples of use, focused on the application of in situ attenuated total reflection (ATR) IR, Raman and UV/vis spectroscopy. The first example shows how the coupling of in situ spectroscopic techniques reveals information concerning the influence of various parameters in catalyst precursor preparation on structural changes of synthesis intermediates and products. In the second example it is shown how in situ spectroscopies can help to answer questions concerning mechanistic aspects arising from a specific type of catalytic reaction.
Experimental aspects
The crucial point in realising a multiple technique approach for studying reactions in the liquid phase is achieving an appropriate setup which allows monitoring of the catalytic system under realistic conditions. Because of their flexibility and comparable ease of handling, the utilisation of spectroscopic probes is generally straightforward for in situ spectroscopic studies of liquid and gas/liquid systems. Since quartz glass is transparent for both Raman excitation lasers and signals and UV/vis rays, the application of these spectroscopies is very versatile. Thus, the respective UV/vis probe and the Raman excitation laser optics can also be focused from outside onto the glass or quartz reaction vessels. In contrast, IR radiation is strongly adsorbed by quartz, hence special infrared transmissive optical fibres are required. Nowadays, silver halogenide fibres have proven suitable and are well established and used for the manufacturing of appropriate ATR-IR immersion probes working in the mid-infrared region, which can be used, for example, in conjunction with conventional Fourier transform infrared (FT-IR) spectrometers via appropriate coupled optics.
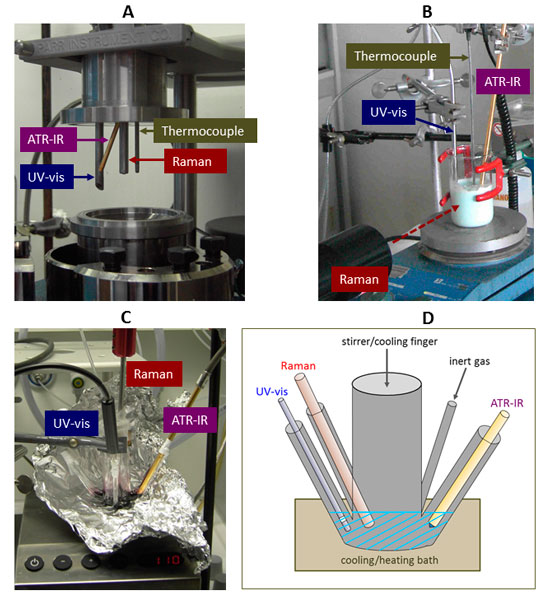
Figure 1. Different setups with implemented fibre-optic probes used for reaction monitoring in liquid-phase systems. A: pressure reactions in an autoclave, B: catalyst preparation, C, D: homogeneously catalysed reaction in a special designed analytical reaction cell.
Typical setups used for the monitoring of liquid-phase reactions are shown in Figure 1: implementation of different probes in the top cover of a modified autoclave4 used for investigations of heterogeneously catalysed liquid phase reactions under pressures up to 20 bar (A), and immersed in an aqueous slurry phase formed during preparation of a mixed metal molybdate catalyst precursor (B). In the latter case, the Raman excitation laser beam was focused from outside.
Because in homogeneous catalysis, there are often several restrictions concerning catalyst and reactant concentrations (the used substrates or ligands may be expensive or merely low amounts are available), a special design for an appropriate analytical reaction cell was developed (see Figures 1 B, C) enabling the simultaneous registration of ATR-IR, UV/vis and Raman spectra from a small volume (5–15 mL) of reaction solution using fibre-optic probes for each spectroscopy.3 This reaction cell can be placed into a heating or cooling bath, and the reaction solution can be stirred using an internal or external stirrer. This cell has no special fittings, thus, making it versatile for different applications. However, its usage is restricted to ambient pressure and temperatures from –70°C to 100°C which are, however, usual conditions for several types of homogeneously catalysed reactions.
For recording the UV/vis spectra, an AvaSpec 2048 fibre optical spectrometer equipped with an AvaLight-DHS light source and a special transmission or reflection probe (Avantes, Apeldoorn, The Netherlands) was used. Raman spectra were recorded using a RXN1-Raman spectrometer (Kaiser Optical Systems) equipped with a 10–400 mW diode laser for excitation at a wavelength of 785 nm and a Mk II Filtered Probe Head with an immersion optic or transmission optic (178 mm focal distance for the laser optical part and the sample). Mid-infrared ATR-IR spectra were measured using a Nicolet Avatar 370 FT-IR spectrometer (Thermo Electron) equipped with a fibre optical diamond immersion probe (Infrared Fiber Sensors, Aachen, Germany).
Synthesis of metal molybdate catalyst precursors in aqueous solution
Heterogeneous molybdenum-based mixed oxides are versatile catalysts for the selective oxidation of alkanes and olefins to their corresponding aldehydes, anhydrides or acids. Their synthesis frequently comprises a series of subsequent steps such as precipitation of precursors, isolation of the precipitate, drying and final calcination. Tailoring such multi-step synthesis protocols for the reproducible and preferential formation of target phases requires a careful control of the different synthesis steps, which may be facilitated by spectroscopic on-line monitoring. The benefit of the latter is demonstrated by the preparation process of a phosphorous-containing iron molybdate precursor.1 The synthesis procedure comprises three consecutive stages: mixing of ammonium heptamolybdate (AHM) and iron nitrate solution (Fe) at 20°C under stirring for 60 min (I), addition of diammonium hydrogenphosphate (DAHP) solution followed by further stirring for 30 min (II), and finally heating and stirring of the slurry for 60 min at 50°C (III). The molar ratio Mo : Fe : P was 12 : 1 : 1.
Raman spectra recorded during stage I show, in addition to the nitrate band, several other bands which are characteristic for the Anderson-type phase A containing [H6FeMo6O24]3– anions (see Figure 2). The additional band at 967 cm–1, rising with time, might stem from a new molybdate phase B. Considering the fact that, depending on the condensation degree of the molybdate ions, the position of the characteristic ns(Mo–Ot) band shifts to higher wavenumbers, phase B should consist of [Mo8O26]4– species, which possibly contain Fe cations. After adding DAHP in stage II, this band immediately vanishes, since phase B dissolves due to the increasing pH. During treatment at 50°C the band intensities strongly increase with time, and the bands at 957 cm–1 and 898 cm–1 shift to 964 cm–1 and 893 cm–1, respectively. This suggests that the nature of molybdate species does not change much, and the observed band shift may be caused by the incorporation of phosphate (phase C).
ATR spectra provide additional information concerning the nature of phosphate species (see Figure 2).
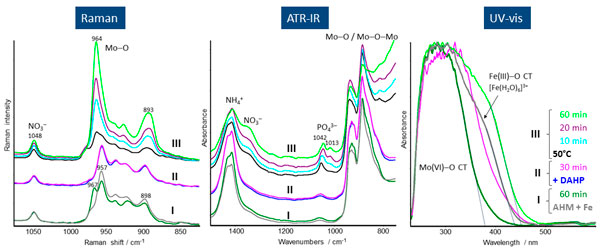
Figure 2. In situ Raman, ATR-IR and UV/vis spectra from a mixture of AHM and Fe nitrate (I), after subsequent DAHP admixture at 25°C (II) and heating to 50°C (III), recorded simultaneously in dependence on time.
After adding DAHP in stage II, a phosphate band appears at 1052 cm–1 superimposed on the initial bands at 1057 cm–1 arising from Fe–OH deformation vibrations. With increasing time at 50°C (stage III), a new phosphate band appears at 1013 cm–1 while the other shifts to lower wavenumbers and becomes more intense with time. Taking account of the Raman spectroscopic results, the 1013 cm–1 band might arise from a new mixed molybdatophosphate (phase C), that possibly coexists with the Anderson phase A in stage III.
The UV/vis spectra (Figure 2) were measured in reflection mode. In this case the formed precipitate itself acts as a reflecting element. Hence, the UV/vis spectra provide exclusive information on the precipitation process and the nature of the precipitate. The spectra recorded during stage I are dominated by the Mo(VI)-O charge transfer (CT) band around 300 nm, whereas the shoulder at 380 nm can be assigned to a Fe(III)-O CT transition of [Fe(H2O)6]3+ species. With reaction progress in stage I the absorption edge of the Mo(VI)-O CT band shifts slightly to lower wavelength, and the Fe(III)-O CT band loses intensity. Both findings are obviously related to the incorporation of Fe into the initial heptamolybdate phase as also suggested by the Raman spectroscopic results. The addition of DAHP in stage II followed by thermal treatment in stage III leads to a strong red shift of the Mo(VI)-O CT absorption edge which indicates the incorporation of phosphate into the molybdate structure as also indicated by ATR-IR and Raman spectroscopy.
Iron catalysed decomposition of formic acid
Formic acid (FA) has attracted attention as a suitable hydrogen storage material. If CO2 is used as the C1-source for the synthesis of FA, a sustainable and reversible energy storage cycle is imaginable. Recently, a well-defined iron catalyst system was described enabling the decomposition of FA to H2 and CO2 in acidic media at ambient conditions with catalyst activities comparable to the best known noble metal catalysts.5 This iron-based catalyst is formed in situ from iron tetrafluoroborate (Fe) and the tetradentate ligand P(CH2CH2PPh2)3 (PP3) in propylene carbonate or tetrahydrofuran (THF) as solvent. However, it was also found that the in situ generated catalyst system Fe/PP3 is sensitive towards traces of chloride leading to a drastic drop in activity depending on chloride concentration. Hence, two aspects were of interest: i) which intermediate complexes were formed by interaction of the iron catalyst complex with FA, and ii) in which way the chloride admixture affects this complex formation. To study these complex formation processes, a comprehensive in situ spectroscopic study comprising simultaneous Raman, UV/vis and ATR-IR spectroscopy was carried out.
The UV/vis spectrum of Fe/PP3/THF (Figure 3A) shows two characteristic bands at 277 nm and 508 nm resulting from p®p* and metal-to-ligand charge transfer (MLCT) transitions, respectively. The observed band at 318 nm results from an interaction between the Fe/PP3 complex and THF as additional ligand, as could be proved by separate experiments. When FA is added to the Fe/PP3/THF solution, this band immediately vanishes and the MLCT band at 508 nm loses intensity and shifts to 499 nm. This finding reveals the coordination of FA on the in situ generated Fe/PP3 catalyst system by replacing previously coordinated THF. After addition of NaCl, new bands at 327 nm and 380 nm appear with increasing intensity in dependence on time, and, simultaneously, the MLCT band shifts continuously to higher wavelengths. Both findings clearly point to a participation of chloride in complex formation. This conclusion could be confirmed by measuring the UV/vis spectrum of the separately synthesised compound [FeCl(PP3)]BF4 in THF which shows a similar spectrum as measured in the in situ experiment after 12 min.
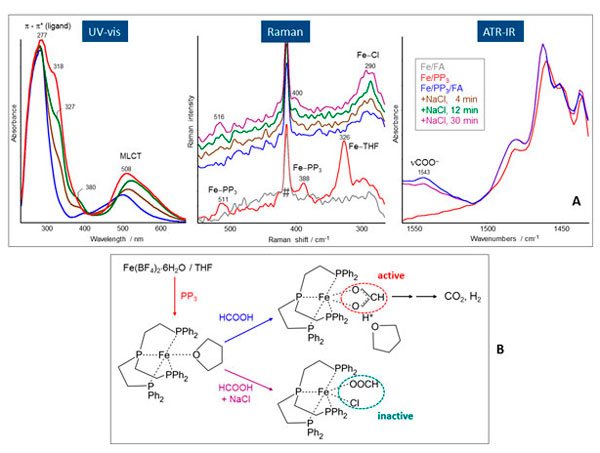
Figure 3. A: In situ UV/vis, Raman and ATR-IR spectra obtained from Fe/PP3 in THF, after addition of FA, and subsequent admixture of NaCl after 4, 12, 30 min. The band from the Raman probe itself is marked by a hashmark (#). B: Scheme of the possible iron complexes generated in the system Fe/PP3/FA/NaCl, and identified by the different spectroscopies.
An inspection of the low wavenumber range in the Raman spectra (Figure 3B) enables information concerning specific features which are characteristic for Fe-ligand and Fe-Cl vibrations. Comparing the spectra of Fe/PP3 (red) and Fe/FA (grey) it is evident that the bands at 511 cm–1, 388 cm–1 and 326 cm–1 appear only in the presence of PP3. While the first two bands result from Fe-PP3 modes in the complex, the latter is related to a Fe-O mode from coordinated THF because in metal aqua complexes metal oxygen modes were observed between 300 cm–1 and 400 cm–1. After addition of FA, the bands at 511 cm–1 and 388 cm–1 disappear first, caused by the diluting effect of FA admixture, but reappear after addition of NaCl, while the intense band at 326 cm–1 does not reappear during the experiment because the former coordinated THF is completely replaced by coordinatively bonded FA (vide infra). This conclusion is in accordance with the UV/vis spectroscopic results revealing changes of the coordination sphere (vanishing of the band at 318 cm–1) by addition of FA. After subsequent addition of chloride, a new band is observable at 290 cm–1, the intensity of which increases with time. This band can be assigned to a Fe-Cl vibration, and reveals the additional coordination of chloride to the Fe/PP3 complex which is in accordance with the UV/vis results.
Concerning the nature of FA coordination, the inspection of the ATR-IR spectra is helpful. In the spectrum of the Fe/PP3/THF a band at 1543 cm–1 is observable (Figure 3C) which was neither detected in FA/THF nor in Fe/FA/THF solutions, merely in the presence of PP3. That means this band only appears when the Fe–PP3 complex was already formed. The position of this band indicates chelating bidentate formate species formed by the coordination of FA on the Fe–PP3 complex. The formation of chelating bidentate formate species was also observed using separately performed FT-IR measurements in transmission mode and simultaneous gas (H2 + CO2) analytics.5 Additionally, it was found that the evolution of CO2 and H2 goes along with formation of this Fe–PP3-bidentate formate complex. However, the admixture of NaCl leads to a distinct drop of catalytic activity depending on the NaCl concentration accompanied by a continuous decrease in intensity of the formate band at 1543 cm–1.
Putting together the specific information provided by each spectroscopic method, it was possible to elucidate the formation process of the active iron complex in the system Fe/PP3/FA and its transformation into the inactive one by chloride admixture (Figure 3B). Thus, this comprehensive in situ spectroscopic study nicely demonstrates the benefit of method coupling.
Conclusions
In situ spectroscopic methods like ATR-IR, Raman and UV/vis spectroscopy proved to be a powerful tools to study the influence of various parameters in preparation processes in liquid-phase systems, and to gain insight into reaction mechanisms and catalyst actions in homogeneously catalysed reactions. The discussed examples of use comprise only a narrow field of application possibilities, but it was the intended aim to demonstrate the variability of in situ spectroscopic methods practiced by the use of fibre-optic probes for each spectroscopy.
A practical problem by combining the different spectroscopies in one experiment is sometimes the covering of different concentration levels, because complementary ATR-IR and Raman spectroscopy need rather higher reactant concentrations compared with UV/vis spectroscopy which requires lower concentrations for getting proper and evaluable spectra. In such cases additional experiments are necessary.
In summary it can be stated that in situ spectroscopic methods and combinations of them offer a great potential for the real-time monitoring of reactions in the liquid phase, for mechanistic studies as well as process control and kinetics.
References
- U. Bentrup, “Combining in situ characterization methods in one set-up: looking with more eyes into the intricate chemistry of the synthesis and working of heterogeneous catalysts”, Chem. Soc. Rev. 39, 4718 (2010). doi: http://dx.doi.org/10.1039/b919711g
- L.R. Knöpke and U. Bentrup, in Heterogeneous Catalysts for Clean Technology-Spectroscopy, Design, and Monitoring, Ed by K. Wilson and A.F. Lee. Wiley-VCH, Weinheim, p. 39 (2014).
- K. Grabow and U. Bentrup, “Homogeneous catalytic processes monitored by combined in situ ATR-IR, UV–Vis, and Raman spectroscopy”, ACS Catalysis 4, 2153 (2014). doi: http://dx.doi.org/10.1021/cs500363n
- L.R. Knöpke, N. Nemati, A. Köckritz, A. Brückner and U. Bentrup, “Reaction monitoring of heterogeneously catalyzed hydrogenation of imines by coupled ATR-FTIR, UV/Vis, and Raman spectroscopy”, ChemCatChem. 2, 273 (2010). doi: http://dx.doi.org/10.1002/cctc.200900273
- D. Mellmann, E. Barsch, M. Bauer, K. Grabow, A. Boddien, A. Kammer, P. Sponholz, U. Bentrup, R. Jackstell, H. Junge, G. Laurenczy, R. Ludwig and M. Beller, “Base-free non-noble-metal-catalyzed hydrogen generation from formic acid: scope and mechanistic insights”, Chem. Eur. J. 20, 13589 (2014). doi: http://dx.doi.org/10.1002/chem.201403602