Rhiannon E. Boseley,a Daryl L. Howard,b Jitraporn Vongsvivut,b Mark J. Hackett*,a and Simon W. Lewis*,a
aSchool of Molecular and Life Sciences, Curtin University, GPO Box U1987, Perth, Australia
bANSTO, Australian Synchrotron, 800 Blackburn Road, Clayton, Victoria, Australia
DOI: https://doi.org/10.1255/sew.2022.a8
© 2022 The Authors
Published under a Creative Commons BY-NC licence
In forensic science, fingerprints are used to connect an individual to a criminal investigation. Their evidential value is reliant on the successful recovery of a fingerprint from a crime scene. Despite a multitude of chemical and physical methods capable of detecting fingerprint residues, there are substantial challenges with fingerprint recovery due to the inherent variability of this biological material. Ongoing research has focused on the development of new or improvement of current fingerprint recovery techniques. Meanwhile, conducting fundamental studies can be valuable to explore the residue itself, to better understand its variation in response to fingerprint treatments.
Spectroscopic methods have played a critical role in the analysis of fingerprints, used to identify the chemical constituents present, examine their degradation over time and compare the chemical variation between donors. Whilst bulk chemical methods have provided an extensive view of the chemical species present, the heterogeneous nature of fingerprint residues means that a more accurate picture can be obtained when the spatial information is preserved. Chemical imaging methods such as mass spectral imaging and infrared micro-spectroscopy can capture the most comprehensive view of fingerprint chemistry.
Recent scientific advances have made spatially resolved chemical analyses more accessible. Among these powerful methods that can provide an unprecedented view of fingerprint chemistry, only a small number of techniques can analyse natural fingerprint samples without any chemical alteration, treatments or solvent extractions prior to analysis. To capture the most realistic representation of the chemical species naturally present in fingerprint residues, in situ, label-free measurements are required. Further, when analysing biological materials, the capability to analyse samples in situ, at ambient temperature and pressure, without the need for vacuum conditions, is important for the stability of loosely bound elemental content and the morphology of the sample.
Fourier transform infrared spectroscopy
Fourier transform infrared (FT-IR) spectroscopy has long been used to characterise unknown samples based on the absorption of IR light by characteristic functional groups of organic molecules. The integration of FT-IR spectroscopy with microscopy (known as FT-IR micro-spectroscopy) further enables classes of biological molecules to be mapped across sample surfaces. Unfortunately, the long wavelengths of IR light (relative to visible light) result in poorer spatial resolution associated with FT-IR micro-spectroscopy than what is generally expected from conventional light microscopy. Nevertheless, the use of high refractive index optical materials in combination with attenuated total reflectance (ATR) modalities, now enable FT-IR micro-spectroscopy to approach micron-scale spatial resolution.1–5
FT-IR micro-spectroscopy has been used not only to investigate how the variation in molecular chemistry can be indicative of donor traits, such as age and biological gender, but also to estimate time since fingerprint deposition.4,6–8 The identification of components correlating to glandular secretions from the eccrine and sebaceous glands has been characterised and their persistence monitored with variables including temperature and time.5 Commonly, fingerprint samples have been analysed with the addition of microscopy (either mapping or imaging), to retain spatial information, highlighting the inherent heterogeneous distribution of organic materials across a sample. In particular, coupling a synchrotron light source to a FT-IR imaging instrument provides enhanced spectral quality and improved data collection times. Dorakumbura et al. exploited the capabilities of synchrotron FT-IR imaging coupled with the ATR attachment to characterise eccrine and sebaceous material within individual droplets in fingerprint residue.9 This method was subsequently applied to monitor the spatio–temporal changes in fingerprint droplets under ambient temperature conditions. Changes in the morphology and chemical composition of the droplet are shown in the immediate hours following deposition (Figure 1), providing a unique perspective of the dynamic nature of this material.10
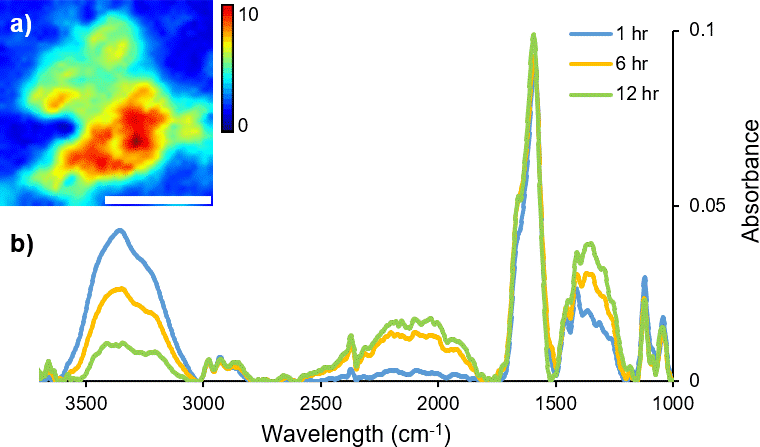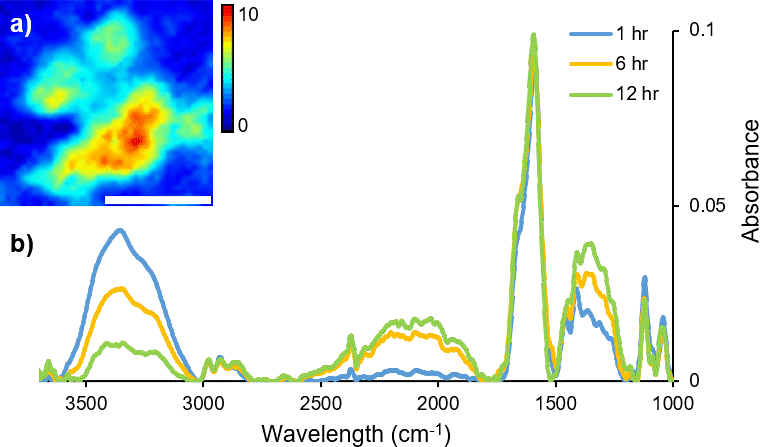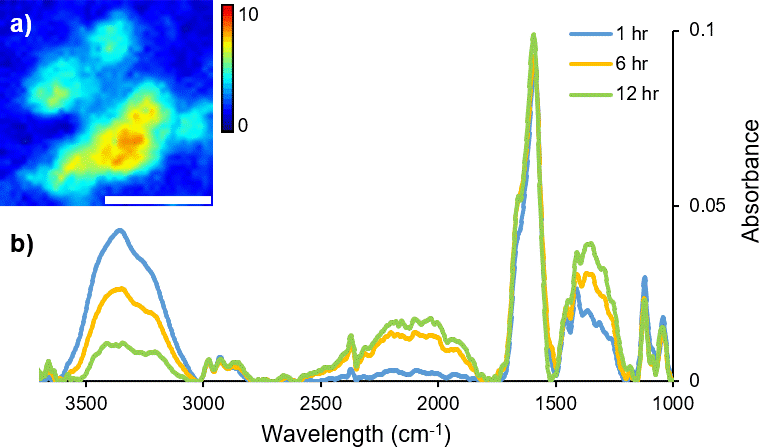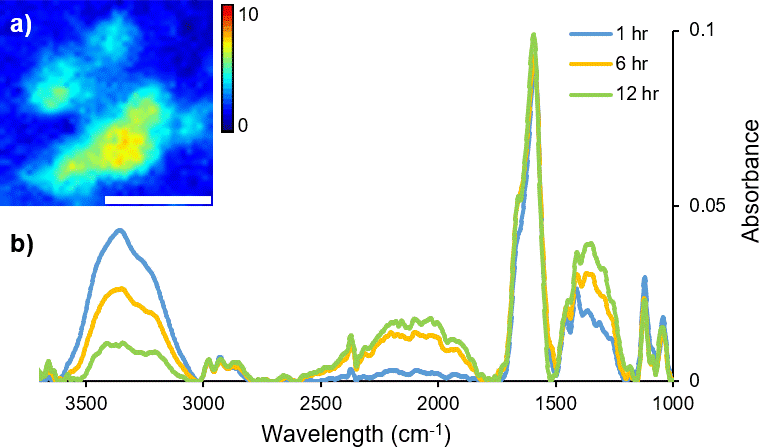
Figure 1. A time-course (12 hour) study of changes in H2O content during air-drying of a droplet within a natural fingermark, as revealed by synchrotron ATR-FT-IR mapping technique. (a) False colour ATR-FT-IR maps were generated by integrating over the ν(O–H) stretching bands of H2O (3000–3500 cm–1) as a marker for eccrine material. Scale bar 20 μm. (b) Representative synchrotron ATR-FT-IR spectra collected from a droplet within a natural fingermark after 1 hour, 6 hours and 12 hours of air-drying.
X-ray fluorescence microscopy
Research into the inorganic material in latent fingerprints has been limited due to the lack of availability in instrumentation with the appropriate sensitivity and spatial resolution required to detect the trace metals present in fingerprint residues. The process of X-ray fluorescence (XRF) inherently lends itself to simultaneous multi-element mapping, which is of great value across many research applications. Coupling of XRF instrumentation with bright X-ray sources (e.g. synchrotron light sources) now provides the opportunity for rapid acquisition of trace-element maps, at micron spatial resolution.11 Recent work conducted by our group has taken advantage of the brightness of synchrotron-sourced X-rays, using X-ray fluorescence microscopy (XFM) to image the distribution of metals and metal ions in natural fingerprints.12 Like its organic counterpart, inorganic material is donor dependent, with great variation in the amount and distribution of elemental material present. The elemental material appeared to follow the ridge pattern detail of fingerprint residue, implying it could be an appropriate chemical target for novel fingerprint development methods. Interestingly, donor behaviour, specifically cosmetic use and contact with metal objects can influence the chemistry of inorganic material in fingerprint residues.13 This information can be exploited for forensic purposes, with metal profiles characterised to particular criminal activities.
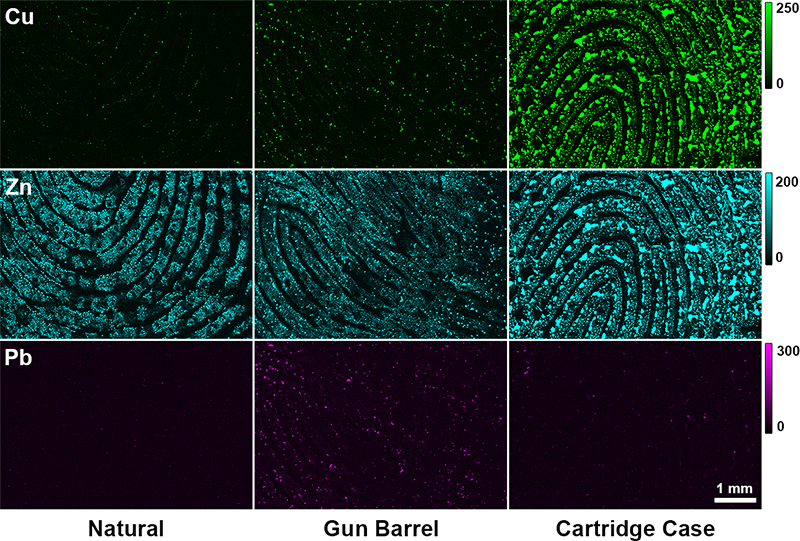
Figure 2. XFM elemental maps from copper, zinc and lead from fingerprints taken following regular activity (left), handling a gun barrel for 30 s (middle) and handling an ammunition cartridge case for 30 s (right). Intensity units are ng cm–2.
Multi-modal chemical imaging studies
To gain a more holistic view of fingerprint chemistry, a multi-modal workflow combining chemical imaging methods was used to investigate the interactions between organic and inorganic materials.12 The same fingerprint sample was imaged using infrared micro-spectroscopy (IRM) and XFM, which together allowed the co-location of metals and metal ions within the organic secretions. Figure 3 shows an example of the metal ions, specifically zinc, located within the organic secretions. A broader distribution of exogenous metal ions was more likely to be linked with external sources such as cosmetic use or secondary metal transfer.12 This finding is critical to understanding how metal ions can interact with chemical treatments, some of which interact with the organic materials, particularly amino acids present within eccrine sweat.

Figure 3. A natural fingerprint deposited on silicon nitride imaged with an optical microscope, XFM elemental mapping showing the zinc distribution, and FT-IR imaging (IRM) showing the distribution of lipid material (false colour image generated by integrating over the C–H stretching band for lipid within the 2800–3000 cm–1 spectral range).
References
- K.L.A. Chan and S.G. Kazarian, “New opportunities in micro- and macro-attenuated total reflection infrared spectroscopic imaging: spatial resolution and sampling versatility”, Appl. Spectrosc. 57(4), 381–389 (2003). https://doi.org/10.1366/00037020360625907
- K.L.A. Chan, S.G. Kazarian, A. Mavraki and D.R. Williams, “Fourier transform infrared imaging of human hair with a high spatial resolution without the use of a synchrotron”, Appl. Spectrosc. 59(2), 149–155 (2005). https://doi.org/10.1366/0003702053085070
- J. Vongsvivut, D. Pérez-Guaita, B.R. Wood, P. Heraud, K. Khambatta, D. Hartnell, M.J. Hackett and M.J. Tobin, “Synchrotron macro ATR-FTIR microspectroscopy for high-resolution chemical mapping of single cells”, Analyst 144, 3226–3238 (2019). https://doi.org/10.1039/C8AN01543K
- A.V. Ewing and S.G. Kazarian, “Infrared spectroscopy and spectroscopic imaging in forensic science”, Analyst 142, 257–272 (2017). https://doi.org/10.1039/C6AN02244H
- C. Ricci, P. Phiriyavityopas, N. Curum, K.L.A. Chan, S. Jickells and S.G. Kazarian, “Chemical imaging of latent fingerprint residues”, Appl. Spectrosc. 61(5), 514–522 (2007). https://doi.org/10.1366/000370207780807849
- D.K. Williams, R.L. Schwartz and E.G. Bartick, “Analysis of latent fingerprint deposits by infrared microspectroscopy”, Appl. Spectrosc. 58(3), 313–316 (2004). https://doi.org/10.1366/000370204322886663
- A. Girod, L. Xiao, B. Reedy, C. Roux and C. Weyermann, “Fingermark initial composition and aging using Fourier transform infrared microscopy (μ-FTIR)”, Forensic Sci. Int. 254, 185–196 (2015). https://doi.org/10.1016/j.forsciint.2015.07.022
- K.M. Antoine, S. Mortazavi, A.D. Miller and L.M. Miller, “Chemical differences are observed in children’s versus adults’ latent fingerprints as a function of time”, J. Forensic Sci. 55, 513–518 (2010). https://doi.org/10.1111/j.1556-4029.2009.01262.x
- B.N. Dorakumbura, R.E. Boseley, T. Becker, D.E. Martin, A. Richter, M.J. Tobin, W. van Bronswjik, J. Vongsvivut, M.J. Hackett and S.W. Lewis, “Revealing the spatial distribution of chemical species within latent fingermarks using vibrational spectroscopy”, Analyst 143, 4027–4039 (2018). https://doi.org/10.1039/C7AN01615H
- R.E. Boseley, J. Vongsvivut, D. Appadoo, M.J. Hackett and S.W. Lewis, “Monitoring the chemical changes in fingermark residue over time using synchrotron infrared spectroscopy”, Analyst 147, 799–810 (2022). https://doi.org/10.1039/D1AN02293H
- D.L. Howard, M.D. de Jonge, N. Afshar, C.G. Ryan, R. Kirkham, J. Reinhardt, C.M. Kewish, J. McKinlay, A. Walsh, J. Divitcos, N. Basten, L. Adamson, T. Fiala, L. Sammut and D.J. Paterson, “The XFM beamline at the Australian Synchrotron”, J. Synchrotron Radiat. 27, 1447–1458 (2020). https://doi.org/10.1107/S1600577520010152
- R.E. Boseley, B.N. Dorakumbura, D.L. Howard, M.D. de Jonge, M.J. Tobin, J. Vongsvivut, T.T.M. Ho, W. van Bronswijk, M.J. Hackett and S.W. Lewis, “Revealing the elemental distribution within latent fingermarks using synchrotron sourced X-ray fluorescence microscopy”, Anal. Chem. 91, 10622–10630 (2019). https://doi.org/10.1021/acs.analchem.9b01843
- R.E. Boseley, D.L. Howard, M.J. Hackett and S.W. Lewis, “The transfer and persistence of metals in latent fingermarks”, Analyst 147, 387–397 (2022). https://doi.org/10.1039/D1AN01951A

Rhiannon Boseley
Rhiannon Boseley is a PhD student within the School of Molecular and Life Sciences at Curtin University, Western Australia. Rhiannon graduated with a BSc Honours (First Class) majoring in Forensic and Analytical Chemistry in 2017 (Curtin University). Her PhD project, titled Using Synchrotron Infrared Spectroscopy and X-ray Fluorescence Microscopy to Explore Fingermark Chemistry, involves fundamental research into the chemical composition of fingermark residue using of a suite of analytical instrumentation at the ANSTO Australian Synchrotron.
[email protected] https://orcid.org/0000-0002-7919-9977
https://orcid.org/0000-0002-7919-9977

Daryl Howard
Daryl Howard is a scientist at ANSTO’s Australian Synchrotron campus. He is applying synchrotron X-ray fluorescence methods to many areas of research including forensics, geology, environmental science and cultural heritage.
[email protected] https://orcid.org/0000-0001-6071-2382
https://orcid.org/0000-0001-6071-2382

Pimm Vongsvivut
Dr Jitraporn (Pimm) Vongsvivut is a Senior Scientist at the Infrared Microspectroscopy (IRM) beamline, ANSTO‒Australian Synchrotron. Her research interest is focused on developments of state-of-the-art vibrational spectroscopy and chemometrics. She has gained extensive experience in the latest development of synchrotron-IR techniques at NSLS (USA), SOLEIL (France), Diamond (UK) and SLRI (Thailand). At ANSTO, she has developed new soft-contact piezo-controlled and in-house macro-ATR techniques widely used in many user experiments. She has published 117 journal articles as primary and co-author in high-impact journals, and has assisted with many experiments and beamline developments as part of her role at the Australian Synchrotron.
[email protected] https://orcid.org/0000-0003-0699-3464
https://orcid.org/0000-0003-0699-3464

Mark Hackett
Dr Mark Hackett is an analytical chemist interested in the development and application of spectroscopic tools to study metal ions in biological systems. Dr Hackett primarily uses X-ray spectroscopies at synchrotron light sources, such as X-ray fluorescence and X-ray absorption spectroscopy to determine elemental distribution and speciation, in situ. These analyses are coupled with a multi-modal workflow, incorporating other spectroscopic tools (Fourier transform infrared spectroscopy, Raman spectroscopy, optical microscopy) to help reveal metal homeostasis in the context of a “holistic biochemical picture”. Dr Hackett received his PhD from The University of Sydney (2011), which was followed by post-doctoral fellowships at the University of Saskatchewan, Canada. Dr Hackett is currently an ARC Future Fellow at Curtin University, Western Australia.
[email protected] https://orcid.org/0000-0002-3296-7270
https://orcid.org/0000-0002-3296-7270

Simon Lewis
Professor Simon Lewis leads the Forensic and Analytical Chemistry Research Group at Curtin University, Perth, Western Australia. He obtained his PhD in Analytical Chemistry from the University of Plymouth, UK before taking up a position as a lecturer at Deakin University, Geelong, Australia (1994–2005) where he was involved in developing and coordinating the undergraduate forensic science program. His group’s research is focused on chemical techniques applied to forensic analysis, particularly in relation to chemical trace evidence and latent fingermarks.
[email protected] https://orcid.org/0000-0002-2049-1586
https://orcid.org/0000-0002-2049-1586