Donatella Capitani,a Noemi Proietti,a Anatoly P. Sobolev,a Riccarda Antiochia,b Maurizio Delfini,c Fabio Sciubba,c Alfredo Miccheli,c Flavio Roberto De Salvadord and Luisa Manninab,a,*
aIstituto di Metodologie Chimiche, Laboratorio di Risonanza Magnetica “Annalaura Segre”, CNR, I-00015 Monterotondo, Rome, Italy
bDipartimento di Chimica e Tecnologie del Farmaco, Sapienza Università di Roma, Piazzale Aldo Moro 5, I-00185 Rome, Italy
cDipartimento di Chimica, Sapienza Università di Roma, Piazzale Aldo Moro 5, I-00185 Rome, Italy
dCRA Consiglio per la Ricerca e la Sperimentazione in Agricoltura, Centro di Ricerca per la Frutticoltura, Via Fioranello 5, I-00134 Rome, Italy
Introduction
Fresh fruits are a rich source of vitamins, mineral salts, antioxidants and fibres. Numerous chemical and chemical–physical analytical methods have been used to control quality, authenticity and origin of these products.1 Among these methodologies, the nuclear magnetic resonance-based (NMR-based) metabolomic approach occupies a prominent position, since, as a non-specific high-throughput analytical method, it has the advantage of being capable of detecting signals due to many different classes of compounds (widely ranging in concentration, with different chemical structures and physicochemical properties) in the same measurement. Many applications have been reported in the literature2 that show the potential of the NMR metabolomic approach in fruit investigations. The monitoring of the metabolic profiling of fruits may also be useful for industries which use fruit-derived nutrients in food production. As case study examples, we report here on the NMR study of kiwifruits and peaches.3,4,5
Materials and methods
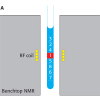
Kiwifruits and peaches were hand-harvested in an experimental field located in the Lazio district, Italy. Rather simple, direct and rapid sample preparation procedures without prerequisite derivatisation of components were applied. Fresh-cut pulp was frozen in liquid N2, finely powdered and submitted to an extraction according to the modified BlighDyer methodology.3,6,7 All NMR spectra of fruit aqueous extracts were recorded at 27°C on a Bruker AVANCE 600 spectrometer operating at the proton frequency of 600.13 MHz. Non-invasive measurements in the field were carried out using a portable unilateral8 NMR instrument from Bruker Biospin.
Results and discussion
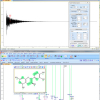
In Figures 1(a) and (b), the 1H NMR spectra of aqueous extracts of kiwifruits and peaches are shown, together with the vertical scale-expansions of aromatic spectral regions. Each fruit type shows a characteristic set of primary metabolites (amino acids, sugars, organic acids) which are almost ubiquitous and some compounds which are specific to the fruit type, which can therefore be considered “markers” of the product. These “markers” are species-specific secondary metabolites usually present in low concentrations; nevertheless, they are often important from the nutritional point of view. The NMR methodology enables primary and secondary metabolites to be identified using 1D and 2D experiments, spiking experiments and utilising literature data.

In the case of kiwifruits, about 40 water-soluble metabolites, including carbohydrates (glucose, sucrose, galactose, fructose, xylose, raffinose, mannose, fructose-6-phosphate and glucose-6-phosphate) and aminoacids (alanine, glutamine, glutamate, threonine, arginine, asparagine, aspartate, valine, leucine, isoleucine, lysine, tryptophane, γ-aminobutyrate, histidine and phenylalanine), organic acids (malic, citric, quinic, ascorbic, lactic, shikimic acid) and other metabolites such as myo-inositol, choline, O3-β-d-glucopyranosyl-trans-caffeic acid, O3-β-d-glucopyranosyl-cis-caffeic acid, adenosine tri-phosphate, uridine, 3-O-α-l-rhamnopyranosylquercetin, neo-chlorogenic acid and epicatechin have been assigned as being present.
In the case of peach fruits, water-soluble metabolites belonging to different classes such as organic acids (citric, fumaric, malic, quinic, shikimic and succinic acid), sugars (fucose, fructose, fructose-6-phosphate, glucose, glucose-6-phosphate, rhamnose, sucrose and xylose), amino acids (alanine, asparagine, isoleucine, threonine, valine and phenylalanine) and other metabolites such as myo-inositol, choline, trigonelline, catechin, chlorogenic and neochlorogenic acids, adenosine tri-phosphate, uridine, orthophosphate and α-l-glycerophosphorylcholine have been identified as being present.
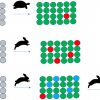
The NMR metabolomic approach, together with suitable statistical analyses, has deepened our knowledge of some important aspects of the investigated foodstuffs. In the case of kiwifruits, the metabolic profiles of aqueous extracts of three different kiwifruit varieties grown in Italy, namely Hayward (Actinidia deliciosa), Zespri Gold (Actinidia chinensis) and CI.GI (a controlled crossbreed from different species of Actinidia deliciosa) kiwifruits have been compared and monitored over a season (from June to December) to investigate the kiwifruit composition of the three varieties at different harvesting times. As expected, in the three cultivars, the content of sugars such as sucrose, glucose and fructose increases during fruit growth, reaching the highest value in December. During the early stages of fruit development, quinic acid represents the highest proportion of the total acids content. In all three cultivars, a wide concentration difference between quinic and shikimic acids is found, the average concentration of quinic acid being about two orders of magnitude higher than that of shikimic acid. In Hayward and CI.GI varieties, shikimic acid disappears at stages of development later than July. In Figure 2, the results obtained from subjecting the NMR data, relative to the three varieties sampled in (a) June and (b) December, to principal component analysis (PCA) are reported. In June, as well as in December, Zespri kiwifruits are always well separated along PC1 from the other two cultivars, whereas Hayward and CI.GI show a separation along PC2 only in June, see Figures 2(a) and (b). This finding is in agreement with the common origin of CI.GI and Hayward as they are both varieties of A. deliciosa. In June, the separation of the Zespri variety from the other two varieties is dominated by two groups of amino acids; in fact, asparagine, glutamine, histidine, triptophane and arginine show higher levels in Zespri than in CI.GI and Hayward, whereas isoleucine and alanine show the opposite behaviour. Fructose shows a higher level in Zespri than in CI.GI and Hayward, whereas sucrose and glucose show the opposite behaviour. Other metabolites such as malic acid and O3-β-d-glucopyranosyl-trans-caffeic acid show higher levels in Zespri than in the other kiwifruits. Hayward and CI.GI immature kiwifruits show the presence of a flavonoid derivative, namely, 3-O-α-l-rhamnopyranosylquercetin that is absent in the Zespri variety. In contrast, Zespri kiwifruits display the presence of neochlorogenic acid, which can be considered a specific marker of this variety. In Hayward and CI.GI kiwifruits, a significant amount of epicatechin is also detected in August5 whereas, in Zespri, the amount of this compound is found to be low over the season. In December, most of the amino acids along with adenosine triphosphate (ATP) and sucrose have a higher level in Zespri than in the other two varieties, whereas glucose and myo-inositol still show a higher level in Hayward and CI.GI than in Zespri fruits.

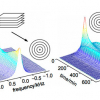
Together with the metabolic profile, the water status of the kiwifruits’ outer pericarp has also been monitored3,4 over the season directly on the intact fruit. Measurements of the T2 spin–spin relaxation time were made by means of a portable unilateral NMR instrument in a fully non-invasive way, see Figure 3(a). The portable NMR instrument allows one to obtain information on the relaxation properties of the kiwifruit texture. As is well known, water compartmentalised in heterogeneous systems, such as foodstuffs, gives rise to multi-exponential decays characterised by multiple time constants. In the three varieties, the two T2 components, namely T2a ascribed to protons in cytoplasm and extracellular space and T2b ascribed to protons in vacuole, become longer over the season. This tendency towards longer T2 relaxation times later in the season is consistent with a change in the fruit texture occurring during fruit development. On average, from October to February, in Hayward and CI.GI T2, relaxation times appear very sensitive to the kiwifruit developmental stage, whereas constant values are measured at times earlier than October. In Zespri kiwifruits, see Figure 3, a net, sharp lengthening of both T2a and T2b occurs between October and November. This behaviour possibly accounts for a sharp transition in the Zespri water state, which would be in agreement with the earlier maturation of the Zespri variety relative to the Hayward and CI.GI varieties.

In the case of peaches, the metabolic profiles of two varieties, Percoca Romagnola 7 and Flaminia, with different susceptibilities to Mediterranean fly (Ceratitis capitata) attack have been compared. Mediterranean fly (medfly) is a species of fly widespread throughout tropical Africa, Australia, the Mediterranean area and in some regions of North, Central and South America. If uncontrolled, the damage due to medfly attack can spread to up to 100% of the crop. The metabolite profiling of peach aqueous extracts analysed using PCA has allowed the comparison between the two varieties. The PCA plot reported in Figure 2(c) shows a clear grouping of the samples according to the variety. The levels of glucose, xylose, myo-inositol, choline, isoleucine and valine are higher in Flaminia than in Percoca Romagnola 7 samples, whereas the levels of fumaric acid, alanine, quinic acid, sucrose, fucose and chlorogenic and neochlorogenic acid are higher in Percoca Romagnola 7 than in Flaminia samples. It is worth noting that the pulp of Percoca, the more resistant variety, presents greater amounts of alanine, quinic acid, chlorogenic and neochlorogenic acid than Flamina. These compounds are reported to be related to the defence against fungal and insect attacks in other members of the Rosaceae family,9 suggesting that the phenylpropanoid pathway is at least partially involved in the repulsion of C. capitata.
Conclusion
The NMR results discussed here show that NMR methodologies play an important role in fruit characterisation as they yield a comprehensive metabolic profile of the analysed samples, provide direct structural information regarding individual metabolites in the mixture and also give information regarding the water state in the tissue. NMR-based metabolic profiling may be a useful tool in discriminating various fruits varieties, in investigating nutritional properties, in monitoring the development of the fruit and in assessing the optimum harvesting time.
References
- S.S. Nielsen, Food Analysis. Springer, New York, USA (2010). doi: 10.1007/978-1-4419-1478-1
- L. Mannina, A.P. Sobolev and S. Viel, “Liquid state 1H high field NMR in food”, Prog. Nucl. Magn. Reson. Spectrosc. 66, 1 (2012). doi: 10.1016/j.pnmrs.2012.02.001
- D. Capitani, L. Mannina, N. Proietti, A.P. Sobolev, A. Tomassini, A. Miccheli, M.E. Di Cocco, G. Capuani, R. De Salvador and M. Delfini, “Monitoring of metabolic profiling and water status of Hayward kiwifruits by Nuclear Magnetic Resonance”, Talanta 82, 1826 (2010). doi: 10.1016/j.talanta.2010.07.080
- D. Capitani, L. Mannina, N. Proietti, A.P. Sobolev, A. Tomassini, A. Miccheli, M.E. Di Cocco, G. Capuani, F.R. De Salvador and M. Delfini, “Metabolic profiling and outer pericarp water state in Zespri, CI.GI and Hayward kiwifruits”, J. Agric. Food Chem. 61, 1727 (2013). doi: 10.1021/jf3028864
- D. Capitani, A.P. Sobolev, A. Tomassini, F. Sciubba, F.R. De Salvador, L. Mannina and M. Delfini, “Peach Fruit: a metabolic comparative analysis of two varieties with different resistance to insect attacks by NMR spectroscopy”, J. Agric. Food Chem. 61, 1718 (2013). doi: 10.1021/jf303248z
- E.G. Bligh and W.J. Dyer, “A rapid method of total lipid extraction and purification”, Can. J. Biochem. Physiol. 37, 911 (1959). doi: 10.1139/o59-099
- A. Miccheli, T. Aureli, M. Delfini, M.E. Di Cocco, P. Viola, R. Gobetto and F. Conti, “Study on influence of inactivation enzyme techniques and extraction procedures on cerebral phosphorylated metabolite levels by 31P NMR spectroscopy”, Cell. Mol. Biol. 34, 591 (1988).
- F. Casanova, G. Perlo and B. Blümich, Single-Sided NMR. Springer-Verlag, Berlin, Germany, (2011).
- S. Michalek, C. Klebel and D. Treutter, “Stimulation of phenylpropanoid biosynthesis in apple (Malus domestica Borkh.) by abiotic elicitors. Eur. J. Hortic .Sci. 70, 116 (2005).